|
9/28/2023 0 Comments What is entropy in chemistry
History Rudolf Clausius (1822–1888), originator of the concept of entropy This description has been identified as a universal definition of the concept of entropy. Upon John von Neumann's suggestion, Shannon named this entity of missing information in analogous manner to its use in statistical mechanics as entropy, and gave birth to the field of information theory. In 1948, Bell Labs scientist Claude Shannon developed similar statistical concepts of measuring microscopic uncertainty and multiplicity to the problem of random losses of information in telecommunication signals. He thereby introduced the concept of statistical disorder and probability distributions into a new field of thermodynamics, called statistical mechanics, and found the link between the microscopic interactions, which fluctuate about an average configuration, to the macroscopically observable behavior, in form of a simple logarithmic law, with a proportionality constant, the Boltzmann constant, that has become one of the defining universal constants for the modern International System of Units (SI). A consequence of the second law of thermodynamics is that certain processes are irreversible.Īustrian physicist Ludwig Boltzmann explained entropy as the measure of the number of possible microscopic arrangements or states of individual atoms and molecules of a system that comply with the macroscopic condition of the system. As a result, isolated systems evolve toward thermodynamic equilibrium, where the entropy is highest. Įntropy is central to the second law of thermodynamics, which states that the entropy of an isolated system left to spontaneous evolution cannot decrease with time. Referring to microscopic constitution and structure, in 1862, Clausius interpreted the concept as meaning disgregation. He initially described it as transformation-content, in German Verwandlungsinhalt, and later coined the term entropy from a Greek word for transformation. In 1865, German physicist Rudolf Clausius, one of the leading founders of the field of thermodynamics, defined it as the quotient of an infinitesimal amount of heat to the instantaneous temperature. The thermodynamic concept was referred to by Scottish scientist and engineer William Rankine in 1850 with the names thermodynamic function and heat-potential. It has found far-ranging applications in chemistry and physics, in biological systems and their relation to life, in cosmology, economics, sociology, weather science, climate change, and information systems including the transmission of information in telecommunication. 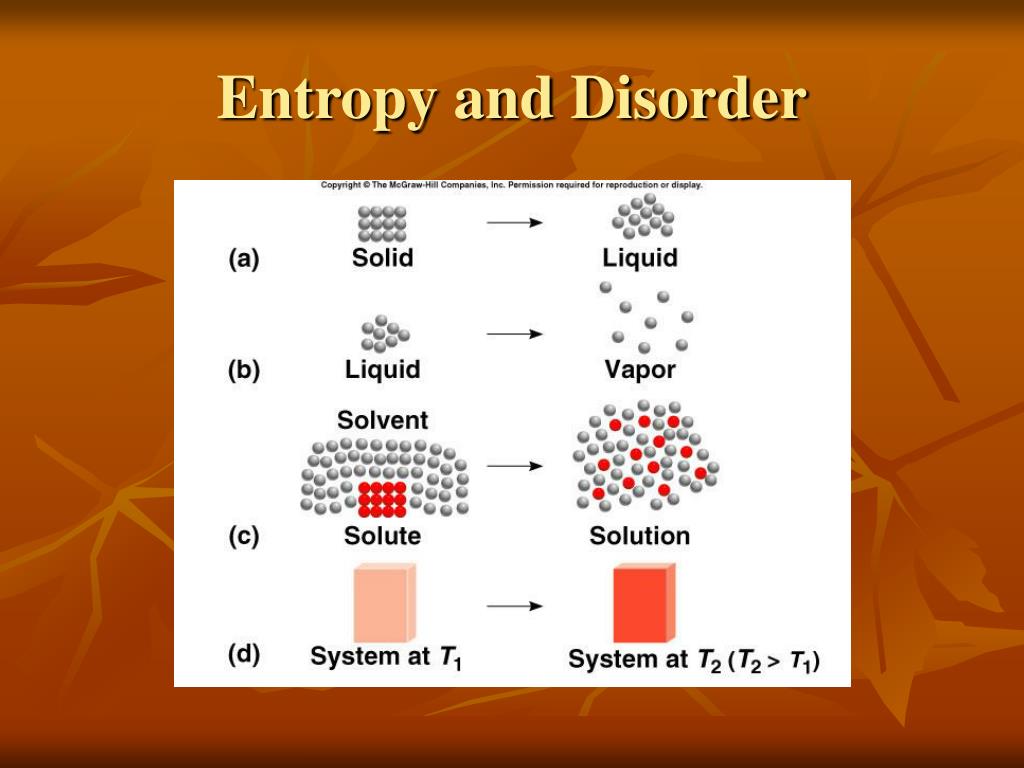 
The term and the concept are used in diverse fields, from classical thermodynamics, where it was first recognized, to the microscopic description of nature in statistical physics, and to the principles of information theory. 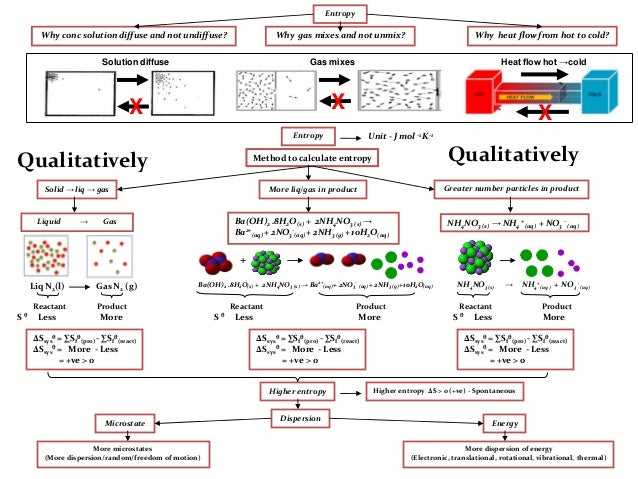
There is nothing in our intuition about order, for example, which suggests that 1 mol Xe gas is more disordered than 1 mol He gas, even though its entropy is in fact larger.Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. It does not lend itself to a quantitative treatment, and it is also difficult to explain some things like the effect of mass in these terms. There are limits to the lengths one can take this order-disorder approach to entropy, though. In other words, W, and hence S, is small for an ordered situation but large for a disordered situation. By contrast, situations which we recognize as disordered, random, or chaotic, can he achieved in a whole variety of ways. A situation which we intuitively recognize as being orderly is also one which can only be achieved in a limited number of ways. This way of looking at entropy is entirely compatible with the approach presented above. In these terms the second law of thermodynamics is seen as a tendency for the disorder of the universe to increase. \)Ī very useful, though somewhat rough, description of the entropy of a substance is as a measure of the randomness or disorder of the atoms and molecules which constitute that substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
